Getting started with KinoML¶
Introduction¶
KinoML is a modular and extensible framework for kinase modelling and machine learning. KinoML can be used to obtain data from online and in-house data sources and to featurize data so that it is ML-readable. KinoML also allows users to easily run ML experiments with KinoML’s implemented models.
In this notebook you will learn how to install KinoML and how to:
Obtain your dataset from ChEMBL
Featurize this data
Train and test a simple Support Vector Classifier (SVC) ML model with your featurized data
And KinoML allows you to do this with just a few lines of code!
For more extensive examples and tutorials have a look at the other notebooks in the KinoML documentation or at the experiments-binding-affnity repository.
The KinoML documentation also allows browsing the API.
Installation¶
KinoML can be easily installed using conda/mamba. We highly encourage using mamba instead of conda to speed up the installation.
mamba create -n kinoml --no-default-packages
mamba env update -n kinoml -f https://raw.githubusercontent.com/openkinome/kinoml/master/devtools/conda-envs/test_env.yaml
conda activate kinoml
pip install https://github.com/openkinome/kinoml/archive/master.tar.gz
Basic example¶
KinoML has a strong focus on kinases, but can be applied to other proteins, if the appropriate code is written. However, the work on kinases is the easiest, since we provide cleaned kinase datasets for ChEMBL and PKIS2, which are hosted at the kinodata repository.
1. Obtaining your data¶
DatasetProvider¶
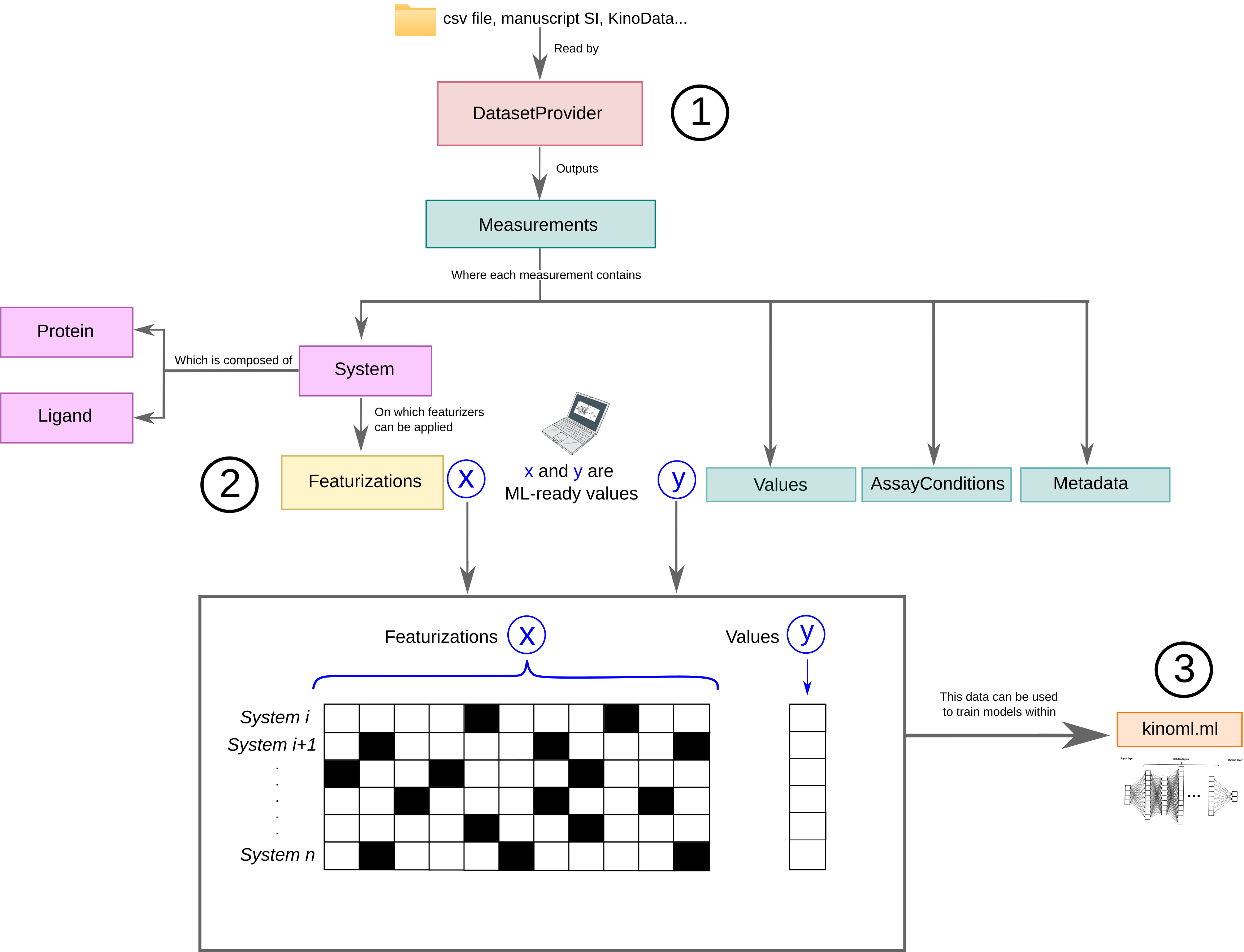
KinoML’s DatasetProvider allows users to easily access and filter datasets. The user can specify the url of the csv file they are interested in or can use the default (as done in the example below). There are different types of DatasetProvider incorporated into KinoML. The one used in this notebook is ChEMBLDatasetProvider, which allows users to use ChEMBL datasets and filter them by specifying the measurement type (“pIC50”, “pKi”, “pKd”) and kinase uniprotID.
DatasetProvider outputs a lists of measurement values, and each of them will be associated to their corresponding system, which is formed by a protein and a ligand. Also, the measurement objects also have Metadata and AssayConditions associated to it, which contains information on how this measurement was obtained.
[1]:
%%capture --no-display
# to hide warnings
from kinoml.datasets.chembl import ChEMBLDatasetProvider
[2]:
chembl_provider = ChEMBLDatasetProvider.from_source(
measurement_types=["pIC50"], #selecting measurement type
uniprot_ids=["P00533"], #kinase of interest
sample=1000, #number of samples you want to work with
)
chembl_provider
[2]:
<ChEMBLDatasetProvider with 1000 measurements (pIC50Measurement=1000), and 986 systems (KLIFSKinase=1, Ligand=986)>
Just looking at the output, we can see that there are 1000 measurements, but only 986 systems. This means there are duplicates in the measurements. Ideally we would delete these duplicated entries, but since this notebook just wants to show a quick example, we will leave this for the other notebooks. Let’s continue and have a look at the first measurement and the stored information.
[3]:
chembl_provider.measurements[0]
[3]:
<pIC50Measurement values=[5.13076828] conditions=<AssayConditions pH=7> system=<ProteinLigandComplex with 2 components (<KLIFSKinase name=P00533>, <Ligand name=OCCCCNc1cncc(-c2cncc(Nc3cccc(Cl)c3)n2)c1>)>>
[4]:
chembl_provider.measurements[0].values
[4]:
array([5.13076828])
[5]:
chembl_provider.measurements[0].system
[5]:
<ProteinLigandComplex with 2 components (<KLIFSKinase name=P00533>, <Ligand name=OCCCCNc1cncc(-c2cncc(Nc3cccc(Cl)c3)n2)c1>)>
[6]:
chembl_provider.measurements[0].system.ligand
[6]:
<Ligand name=OCCCCNc1cncc(-c2cncc(Nc3cccc(Cl)c3)n2)c1>
[7]:
chembl_provider.measurements[0].system.protein
[7]:
<KLIFSKinase name=P00533>
As explained above, each measurement comes with a values array representing the activity values for this measurement, which can be considered the typical y we want to predict in an ML experiment. The system object contains relevant information about protein and ligand for this measurement. The system information is typically X in an ML experiment, but is not yet in a machine-friendly format.
2. Featurize your data¶
Featurizer¶
To get the X (system information) in a machine-friendly format, KinoML uses so called featurizers, which encode the information of each system. KinoML has different featurizers implemented. In this notebook we will use the MorganFingerprintFeaturizer. We are going to iterate this featurizer over all systems and transform the ligand into a bit vector. All performed featurizations are commonly stored in the featurizations attribute of each system. The last performed
featurization is stored additionally for easy access.
[8]:
from kinoml.features.ligand import MorganFingerprintFeaturizer
[9]:
%%capture --no-display
# to hide warnings
chembl_provider.featurize(MorganFingerprintFeaturizer())
chembl_provider.measurements[0].system.featurizations
[9]:
{'last': array([0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0,
0, 0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 1, 0,
0, 1, 0, 0, 0, 0, 0, 0, 0, 1, 1, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 1, 0, 1, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0, 1, 0, 0, 0,
0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 1, 0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0,
0, 1, 0, 0, 0, 1, 0, 0, 0, 0, 0, 1, 0, 0, 1, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 1, 0, 1, 0, 0, 0,
0, 0, 1, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0,
0, 0, 0, 1, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0, 0, 0, 0, 1,
0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 1, 0, 0, 1, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 1, 0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 1, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 1]),
'MorganFingerprintFeaturizer': array([0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0,
0, 0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 1, 0,
0, 1, 0, 0, 0, 0, 0, 0, 0, 1, 1, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 1, 0, 1, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0, 1, 0, 0, 0,
0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 1, 0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0,
0, 1, 0, 0, 0, 1, 0, 0, 0, 0, 0, 1, 0, 0, 1, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 1, 0, 1, 0, 0, 0,
0, 0, 1, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0,
0, 0, 0, 1, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0, 0, 0, 0, 1,
0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 1, 0, 0, 1, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 1, 0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 1, 0, 0, 0, 0,
0, 0, 0, 0, 0, 0, 0, 1, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0, 0,
0, 0, 0, 0, 0, 1])}
For more details on the KinoML object model have a look at the respective notebook.
3. ML model training and testing¶
ML training and testing¶
Great, we have used KinoML to first obtain the data we wanted to work with, and then to featurize this data and make it ML readable. Now that we have X and y, we can run a small ML experiment. In this case, we will train a support vector classifier from sklearn.
[10]:
import matplotlib.pyplot as plt
from sklearn.svm import SVC
from sklearn.metrics import roc_curve, roc_auc_score
from sklearn.model_selection import train_test_split
[11]:
# get data from provider
X, y = chembl_provider.to_numpy()[0]
print(X[0])
print(y[0])
[0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 1 0 0 0 0 0 0 0 0 0 0 0 0 0 1 0 0 0 0 0 0 0
0 0 0 1 0 0 0 0 0 0 0 0 1 0 0 0 0 0 0 0 0 0 0 0 0 0 0 1 0 0 1 0 0 0 0 0 0
0 1 1 0 0 0 1 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 1 0 1 0 0 0 0
0 0 0 0 0 0 0 0 0 0 0 0 0 1 0 0 0 1 0 0 0 0 0 0 0 1 0 0 0 0 0 1 0 0 0 0 1
0 0 0 0 0 0 0 1 0 0 0 1 0 0 0 0 0 1 0 0 1 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0
0 0 0 0 0 0 1 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 1 0 0 1 0 1 0 0 0 0 0
1 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 1 0
0 0 0 0 0 0 0 0 1 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 1
0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 1 0 0 0 0 0 0 0 0 0 1 0 0 0
0 0 0 0 1 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 1 0 0 0 0 0 0 1 0 0 0 0 0 0
0 0 0 0 0 1 0 0 1 0 0 0 0 0 0 0 0 0 0 0 0 0 1 0 0 0 0 0 0 0 0 0 0 0 0 0 0
0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 1 0 0 0 0 0 0 0
0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 1 0 0 0 0 1 0 0 0 0 0 1 0
0 0 0 0 0 0 0 0 0 0 1 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 0 1]
5.1307683
[12]:
# binarize activity values
y = (y > 7).astype(int)
print(y[0])
0
[13]:
# split data into train and test sets
x_train, x_test, y_train, y_test = train_test_split(X, y, test_size=0.2)
[14]:
# train the support vector classifier
svc = SVC(probability=True)
svc.fit(x_train, y_train)
[14]:
SVC(probability=True)
[15]:
# get the ROC curve including AUC
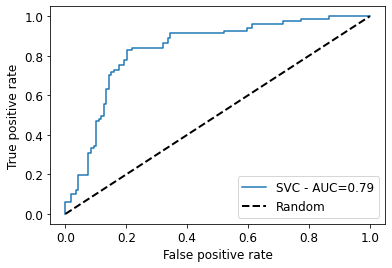
y_test_pred = svc.predict(x_test)
svc_roc_auc = roc_auc_score(y_test, y_test_pred)
fpr, tpr, thresholds = roc_curve(y_test, svc.predict_proba(x_test)[:,1])
plt.plot(fpr, tpr, label=f'SVC - AUC={round(svc_roc_auc,2)}')
plt.xlim([-0.05, 1.05])
plt.ylim([-0.05, 1.05])
plt.plot([0, 1], [0, 1], linestyle='--', label='Random', lw=2, color="black") # Random curve
plt.xlabel('False positive rate', size=12)
plt.ylabel('True positive rate', size=12)
plt.tick_params(labelsize=12)
plt.legend(fontsize=12)
plt.show()
For more advanced examples have a look at the experiments-binding-affinity repository.